Is My Breast Cancer Coming Back?
Explore the critical role of circulating Tumor DNA (ctDNA) in breast cancer surveillance with insights from Dr. Barry Rosen. This guide delves into ctDNA’s impact on monitoring and detecting cancer recurrence, featuring advancements like the groundbreaking Signatera™ technology. Essential for survivors, it offers understanding and reassurance in managing breast cancer’s journey.
If you’ve ever found yourself asking, ‘Is my cancer gone?’ or ‘Is my cancer coming back?’ — you’re not alone. These are questions that resonate deeply with many individuals, including myself, from the moment of diagnosis. Dr. Rosen, Breast Surgical Oncologist, Senior Medical Advisor for Learn Look Locate, often hears these concerns from his patients, understanding the weight they carry. Here, we aim to provide not just answers, but comfort and a sense of community. I hope you find this to page to be a resource in your journey.
– xoxo, Cynthia, Stage 2b, Founder, Learn Look Locate.
What is ctDNA?
Circulating Tumor DNA (ctDNA) is a groundbreaking discovery in the realm of cancer research, offering a beacon of hope for breast cancer survivors. Comprising small fragments of DNA shed by cancer cells into the bloodstream, ctDNA serves as a crucial indicator of the presence or recurrence of cancer. Its significance in breast cancer survivorship lies in its ability to enable more precise and personalized monitoring and treatment strategies.
The advent of ctDNA testing marks a transformative approach in cancer care, allowing for a more proactive and individualized management of the disease.


Lymph Nodes: Sentinels in the Fight Against Breast Cancer
Lymph nodes are pivotal in the battle against breast cancer, acting as sentinels that signal the potential spread of the disease. As cancer cells break away from the primary tumor, they frequently migrate first to the nearby lymph nodes.
Analyzing these nodes is essential for determining the extent of cancer’s spread and forms a critical component of the breast cancer treatment decision process. The status of lymph nodes can profoundly influence the choice of therapies, highlighting their vital role in comprehensive cancer care and management.

Expert Insights: Dr. Barry Rosen on Breast Cancer Management
Dr. Barry Rosen stands at the forefront of breast cancer care, offering a wealth of knowledge and experience that is invaluable in the fight against this disease.
His insights shed light on the multifaceted nature of breast cancer management, encompassing aspects from diagnosis to treatment, and the importance of post-treatment surveillance. Dr. Rosen’s expertise helps demystify the complexities of breast cancer, offering clarity and direction to those navigating this challenging journey.
Watch the exclusive interview for more:
Understanding MRD Testing in Breast Cancer Care
Molecular Residual Disease (MRD) testing is revolutionizing oncology, offering a highly sensitive method to detect the faintest traces of cancer that might linger post-treatment. This advanced testing is crucial for assessing the risk of cancer recurrence and plays an integral role in customizing post-treatment monitoring and care.
MRD testing’s precision in detecting minute cancer cells provides oncologists with a powerful tool to evaluate treatment efficacy and helps in strategizing future therapeutic interventions, ensuring that each patient’s treatment plan is as effective and targeted as possible.


Signatera™: Personalized Surveillance in Breast Cancer Care
Signatera™ represents a significant advancement in breast cancer care. This personalized, tumor-informed ctDNA molecular residual disease (MRD) testing provides critical insights to guide cancer treatment decisions.
Innovative Testing: How Signatera™ Operates
- Tissue and Blood Sample Analysis
- Ongoing Monitoring Through Blood Tests
- Interpreting Changes in ctDNA Levels
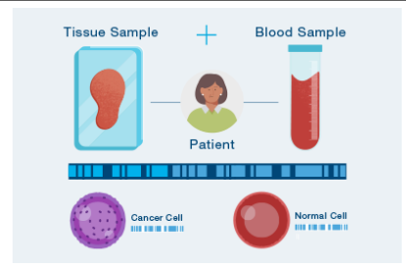
- The journey with Signatera™ begins with a one-time tissue and blood sample, forming the foundation of your unique test. This initial step is vital in tailoring the test to your specific cancer profile.

2. After establishing your baseline, regular blood draws are required as prescribed by your doctor. These tests track changes in your ctDNA levels, offering valuable information about the progression or remission of your cancer.
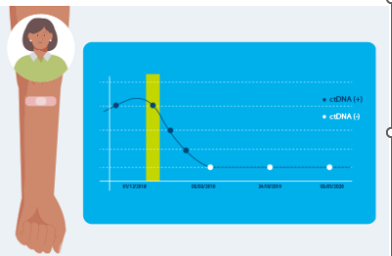
3. Repeated testing with Signatera™ allows for a detailed understanding of how your cancer responds to treatment. This ongoing monitoring is crucial in adapting your treatment plan as needed.
Tailored Care with Signatera™

Detecting Residual Cancer
Post-surgery and initial treatment, Signatera™ aids in determining if any cancer traces remain. This insight is crucial in evaluating recurrence risks and shaping future treatment strategies.

Monitoring for Cancer Recurrence
For those with high-risk breast cancer, early detection of recurrence at a molecular level is possible with Signatera™, potentially even before it’s visible on scans. This early warning system is vital for timely intervention.

Assessing Immunotherapy Effectiveness
Signatera™ also plays a key role in understanding the impact of immunotherapy treatments, tracking ctDNA levels to gauge treatment success.
Real Stories, Real Impact: Survivor Voices
Discover the powerful stories of breast cancer survivors who found hope and direction through Signatera™. Their experiences illuminate the real-world impact of this innovative testing.

“There are few methods available to test for recurrence, at which point there has to be a visible tumor or lesion, yet there are countless ways for your anxiety to spiral every time you get a random headache or pain. The Signatera™ test has been such an empowering addition to my continued monitoring! It’s provided peace of mind against my spiraling anxiety — there’s biological proof that I’m NED — as well as the knowledge that, if I have a recurrence, it will be caught early. Knowledge is not only power, but also reassurance.”
Beth DeLong

“The peace of mind I’ve been getting with Signatera™ is truly priceless. It’s truly amazing I can monitor for the first signs of a recurrence at a molecular level. For this I am deeply grateful, because knowing earlier really does matter in treating and managing the disease. There are always ways to take action when we have the knowledge to empower us. I look forward to continuing to use this test for years to come.”
Kari
FAQs
What is the most challenging aspect of breast cancer recurrence?
The uncertainty of whether cancer cells have left the primary site and spread elsewhere in the body is a significant concern for both patients and doctors.
How does ctDNA testing help in cancer surveillance?
ctDNA testing detects the presence of cancer DNA in the bloodstream, offering a dynamic and sensitive way to monitor for cancer recurrence.
What is the role of lymph nodes in breast cancer?
Lymph nodes are often the first location to which cancer cells spread from the primary tumor. Examining lymph nodes helps determine the extent of cancer spread and is vital in staging the cancer and planning treatment.
What is Molecular Residual Disease (MRD) testing, and how does it relate to ctDNA?
MRD testing is used to detect minute traces of cancer that may remain after treatment. ctDNA is a key component of MRD testing, helping to identify the smallest presence of cancer at a molecular level, which is crucial for assessing recurrence risks.
How does Signatera™ differ from other ctDNA tests?
Signatera™ is a personalized, tumor-informed ctDNA test that’s tailored to each individual’s unique cancer profile. It offers highly specific monitoring for cancer recurrence, providing a more accurate and personalized approach compared to standard ctDNA tests.
Can Signatera™ detect cancer recurrence before it's visible on scans?
Yes, Signatera™ is designed to detect molecular signs of cancer recurrence, potentially identifying it earlier than traditional imaging methods, which can be crucial for early intervention.
How can patients access Signatera™ testing?
Who can benefit from Signatera™?
Signatera™ is particularly beneficial for breast cancer patients, especially those at high risk or undergoing specific treatments like immunotherapy. It is also covered by Medicare for stage IIb and higher breast cancer patients.
What steps should I take if I'm concerned about breast cancer recurrence?
If you’re concerned about recurrence, it’s important to discuss your risk and surveillance options, including ctDNA testing like Signatera™, with your healthcare provider. Regular check-ups and staying informed about new advancements in cancer monitoring are also crucial.
Why Choose Signatera™: A New Era in Cancer Management
Page is sponsored by Signatera™.
Dr. Barry Rosen, Senior Medical Advisor for LLL, asserts, “Signatera™ takes cancer management into the next generation of medicine. It harnesses a patient’s unique tumor mutation signature to provide timely and precise detection of disease recurrence.“
Doctor's Perspective
Barry Rosen, MD, FACS
Senior Medical Advisor, Learn Look Locate
“Signatera™ takes cancer management into the next generation of medicine. It harnesses a patient’s unique tumor mutation signature to provide timely and precise detection of disease recurrence. Simply put, early detection has always been the key to fighting cancer and its recurrence. And reliable surveillance testing is the master key we’ve all been looking for.”

Educate. Inspire. Connect.
Sign up for our newsletter.

